Welcome to our Research page!
Our research is focused on understanding the innate and adaptive immune responses in skin infections and diseases. We study inflammasome biology, caspase signaling, and immune metabolism in inflammatory skin diseases, including S. aureus skin infections, atopic dermatitis, and psoriasis.
With the emergence of antibiotic-resistant bacteria, such as community-acquired methicillin- resistant S. aureus (CA-MRSA), there is an unmet clinical need to develop immune-based therapies to treat skin infections. Therefore, understanding the host immune response to bacterial infections is imperative in identifying novel host-mediated therapies.

As an immune evasive mechanism, S. aureus induces apoptosis in immune cells, including macrophages and monocytes. However, to counter the infection, the host mounts an inflammatory response, predominantly orchestrated by neutrophils and the cytokine IL-1β, which are necessary for the clearance of bacterial infection. In the process of secretion of active IL-1β cytokine, the cells activate inflammasomes.
Inflammasomes are multi-protein complexes assembled upon detecting pathogen or damage-associated signals (PAMPs or DAMPs). Nod-like receptors (NLRs), ASC, and caspases, including Caspase-1/-11 and -8, can combine to form the inflammasome complex detected by ASC speck formation. And once activated and cleaved, the active caspases are essential effectors in the secretion of the cytokine IL-1β.
Our Goal
“To understand the immune responses in skin diseases and to develop biomarkers and potential therapeutic targets.”
We have identified pan-caspase inhibition as a potential host-directed immunotherapy against MRSA and other bacterial skin infections. In pursuit of finding novel therapies, our lab continues to work on multiple projects to understand the effect of pan-caspase inhibition on the adaptive immune responses, and metabolic changes in immune and non-immune cells.
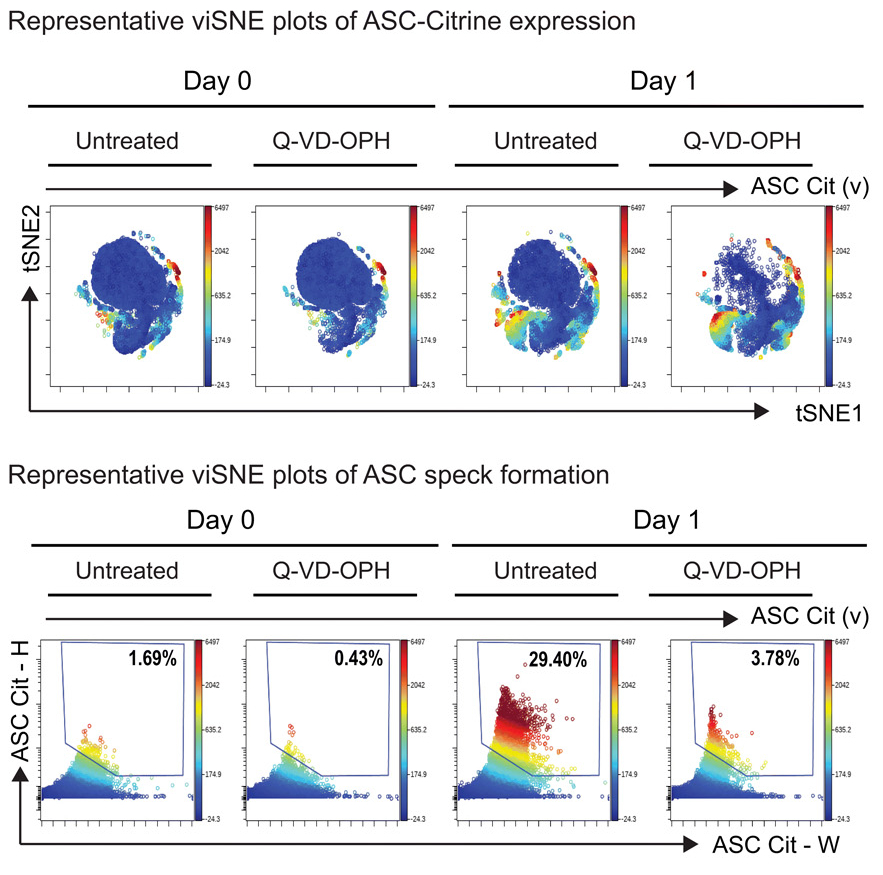
To further understand the inflammasome-mediated mechanisms that regulate host defense we study the role of NLRP12 and Caspase-8 mediated inflammasome signaling during in vivo S. aureus infection.
Taking advantage of bioluminescent bacterial strains and fluorescent in vivo preclinical models, we use imaging techniques to non-invasively and longitudinally track bacterial clearance and host immune responses over time.
We include multi-OMICS, including single-cell high-dimensional Fluorescent Activated Cell Sorting (FACS) and scRNA sequencing techniques and apply machine learning algorithms to decipher the molecular underpinnings of the host response in inflammatory skin diseases.
Understanding the Immunometabolic pathways
Studying the metabolic pathways involved in immune cell function and survival will aid in developing immunotherapies and strategies for combating infectious diseases.


Immune cells use several metabolic pathways to generate adequate energy stores to support their varied functions and survival. During an infection, the host immune cells alter their metabolic programs to facilitate their effector functions, including cytokines secretion. It is becoming increasingly evident that the metabolic program of immune cells modifies the inflammatory status and overall effectiveness in clearing the infection. There is a need to identify new approaches and enhance already established techniques, including fluorescence-activated cell sorting (FACS) based metabolic assay (Met-Flow), to understand the interplay of metabolic pathways in immune cells.
Visit our Publications page!
Contact us to learn more about our work!

