Prologue
The morning was about to dawn and the radiance of the golden hour filled the sky. Swayed by his overwhelming senses, Ma was standing against the waves of the ocean caressing his feet and its sounds permeating his ears. As he turned around, he saw Zen Master walking towards him
“What did you learn?” he asked
“The massive ocean is so overwhelming, I don’t know where to focus!” replied Ma with concern
Understanding the impatience in Ma’s voice, Zen Master with all his mature wisdom, whispered:
“Search! Slowly try to focus on what interests you. The key is finding your interest. Look around, things you see and the one’s you don’t. Stay with it! Make that interesting point your passion. With time, the passion will become so communicable that you will steadily experience the overwhelming ocean with your mind. And in that oneness, pay attention; the ocean will teach you what you have to learn. Remember, the key is finding your interest!”
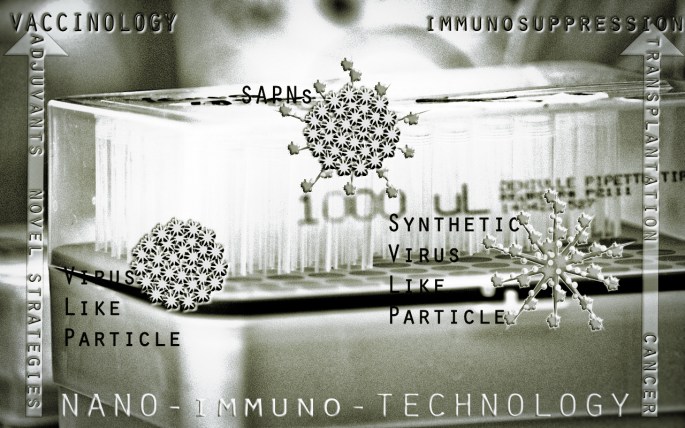
The meeting point of Nantechnology and Immunology
Oceans they are! The discipline of Nanotechnology and Immunology are boundless. Trying to find an interesting point of focus at their confluence is as intriguing as the subject themselves. Therefore, the aim of this short digest is not to cover all the important advancements or possible applications; but rather an attempt to focus at the accent point of the fascinating intersection of both worlds.
 A nanometer (nm) is a unit of measurement that equals to one billionth of a meter. The technology that uses the unique properties of objects within the overall size range of 1-1000 nm is called Nanotechnology. Yes, size does matter! Especially when it comes to immune responses. Bio substances that induce an immune reaction, including antigens or pathogen associated molecular patterns (PAMPS) are in nm scale. The physical and chemical properties of a substance such as size, shape, and hydrophobicity determines its ability to induce an immune response. And the ability to engineer properties of substances towards modulating an immune reaction, fields the application of nanotechnology in Immunology. And what an interesting field it is! The last decade or so has seen an exponential advancement in the field of nanotechnology. Limited by scope, this short summary aims to focus on its application in development of vaccines.
A nanometer (nm) is a unit of measurement that equals to one billionth of a meter. The technology that uses the unique properties of objects within the overall size range of 1-1000 nm is called Nanotechnology. Yes, size does matter! Especially when it comes to immune responses. Bio substances that induce an immune reaction, including antigens or pathogen associated molecular patterns (PAMPS) are in nm scale. The physical and chemical properties of a substance such as size, shape, and hydrophobicity determines its ability to induce an immune response. And the ability to engineer properties of substances towards modulating an immune reaction, fields the application of nanotechnology in Immunology. And what an interesting field it is! The last decade or so has seen an exponential advancement in the field of nanotechnology. Limited by scope, this short summary aims to focus on its application in development of vaccines.
Road to Vaccinology via Nanotechnology
Nanoparticles as adjuvants
The goal of an effective vaccine is to enhance the quality and quantity of the adaptive immune response (both the humoral and cellular). To this effect adjuvants like alum are commonplace in vaccines. An adjuvant is a substance that is added to a vaccine to increase the body’s immune response to the vaccine. Nanoparticles in form of nanoemulsions (oil-in-water emulsions) that are composed of solvents and surfactants provide such adjuvant activity. An example in current use is MF59 (Novartis), which consists of squalene oil (a natural 30 carbon organic compound from shark liver oil or amarnath seeds) in combination with polysorbate 80 and sorbitan trioleate. MF59 is licensed as adjuvant in influenza virus vaccines in Europe under the commercial name FLUAD®. MF59 have been shown to be a more potent adjuvant than alum for inducing humoral and Th1 type cell mediated (cellular) immune response. It is interesting to note that MF59 is yet to be approved in the Americas. Apart from nanoemulsions, nanoparticle based vaccine carriers that are in development as adjuvants include biodegradable poly(lactide-co-glycolide) (PLGA) nanoparticles, co-polymer hygdrogels or ‘nanogels’, cholesterol-bearing hydrophobized pullulan (CHP) and cationic lipsosomes. These nanoparticles raise interest in the field due to their size and immunomodulation properties.
Novel vaccine strategies
Nanoparticles as adjuvants may sound unimaginative. Imagine this! How about engineering nanoparticles as complete vaccines? That should sound novel in approach. Right? Well, such vaccines are in development and in preclinical and close to clinical trials. Although some genuine ethical concerns do swell against these vaccines, the immunological outcome looks promising. Virus like Particles (VLPs) and Self Assembling Protein Nanoparticles (SAPNs) are a two such nanoparticles engineered towards development as complete vaccines.
Virus like Particles (VLPs) based vaccines
With their ability to replicate and undergo genetic recombination, viruses are the smallest among pathogens and yet ingenious ones that find ways to evade our defense mechanisms. Taking cues from viruses, Nanotechnologists have developed virus like particles (VLPs) as novel strategies for vaccines. VLPs unique size, shape, uniformity and stability of structures closely resemble viruses. However, they do not contain genetic material and lack replicative abilities. VLPs are nanoparticles (20-100 nm) that can be broadly divided into two categories, Non-synthetic VLPs and Synthetic VLPs.
Non-synthetic VLPs can be designed to contain viral protein subunits that have the ability to form a viral capsid. Currently, there are about 20-30 different enveloped or non-enveloped VLPs in preclinical and clinical development against infectious diseases and cancer. As a strategy, one of the main advantages of VLPs that could be exploited is incorporation of other biological structures such as Toll Like Receptor (TLR) ligands, cell targeting moieties or other biologically active mediators that could enhance the efficiency of vaccines.
Synthetic VLPs are derived by chemical synthesis of lipopeptide monomers that could enhance nanoparticle assembly and stabilize the three-dimensional conformational structure of protein antigens. One promising example of Synthetic VLPs that is currently in development is Lipopeptide-based synthetic VLPs (20-30 nm), that have been used to repetitively display a peptide-mimetic epitope derived from V3 variable loop of gp120 protein of HIV virus.
Self Assembling Protein Nanoparticles (SAPNs)
Learning from microbial pathogens and VLPs, SAPNs are designed using systemic modeling. By taking advantage of both naturally occurring and synthetically engineered biomolecules, SAPNs are designed to optimize immune responses to vaccine antigens. In essence we could call SAPNs as Version.2 of VLPs. SAPNs can be designed to achieve repetitive antigen display for effective vaccine development. In the process, it can enable conformational presentation of inserted protein epitopes in exposed configuration that could protrude or extend from the surface of the particle after assembly of subunits. About 180 peptide chains can be assembled into a single nanoparticle to form an icosahedral structure. The advantage in this geometry enables better antigen display and cellular activation that could lead to significant increases in the specific production of high-titer, high affinity antibodies directed against the inserted antigen epitopes. A successful example of SAPNs is the incorporation of trimeric coiled-coil epitopes from surface protein of severe acute respiratory syndrome coronavirus (SARS-CoV). These SAPNs has been shown to strongly induce virus-specific neutralizing antibodies in mice after immunization. Similar SAPNs specific to malarial parasite Plasmodium falciparum and P. berghei are in development.
Immunologists consider vaccines as an integral part in prevention and fight aginst infectious diseases. Apart from infectious diseases, advantages of vaccines have found ground in fight against chronic diseases and cancer. At this juncture, application of nanotechnology in form of adjuvants and novel vaccine (VLPs or SAPNs) strategies buoy up the world of vaccinology. Over the long haul, some of the ethical concerns regarding the use of nanoparticles are valid and inflammatory status and long-term effects of these nanoparticles should be extensively studied. Yet, important developments in the field of nanotechnology have already found its application in fields of immunosuppression, transplantation and cancer immunotherapy. Furthermore, exploiting its use in experimental immunology both in vitro and in vivo can provide novel application methods and can potentiate our understanding of our immune system.
Epilogue
The rays of the morning sun warmed Ma, as he was charged with vim and vigour from Zen Master’s counsel.
Little did he know that he had already stumbled upon his point of interest. I should tell this to the Zen Master he thought, but with a smile returned home thinking to himself
“Tomorrow is another day, yet another morning will dawn to teach more. The ocean is full of lessons!”
References:
(1) Smith DM, Simon JK, Baker JR. Applications of nanotechnology for immunology. Nat Rev Immunol. 2013 Jul 25;13(8):592–605.
(2) Torres Andón F, Alonso MJ. Nanomedicine and cancer immunotherapy – targeting immunosuppressive cells. J Drug Target. Informa Healthcare; 2015 Aug;23(7-8):656–71.
(3) Abuelma’atti MT. Nanotechnolgy: Benefits, risks and ethical issues. Exhibition, “Innovative Engineering for Sustainable Environment.” IEEE; 2009. 5 p.